Too much is like not enough
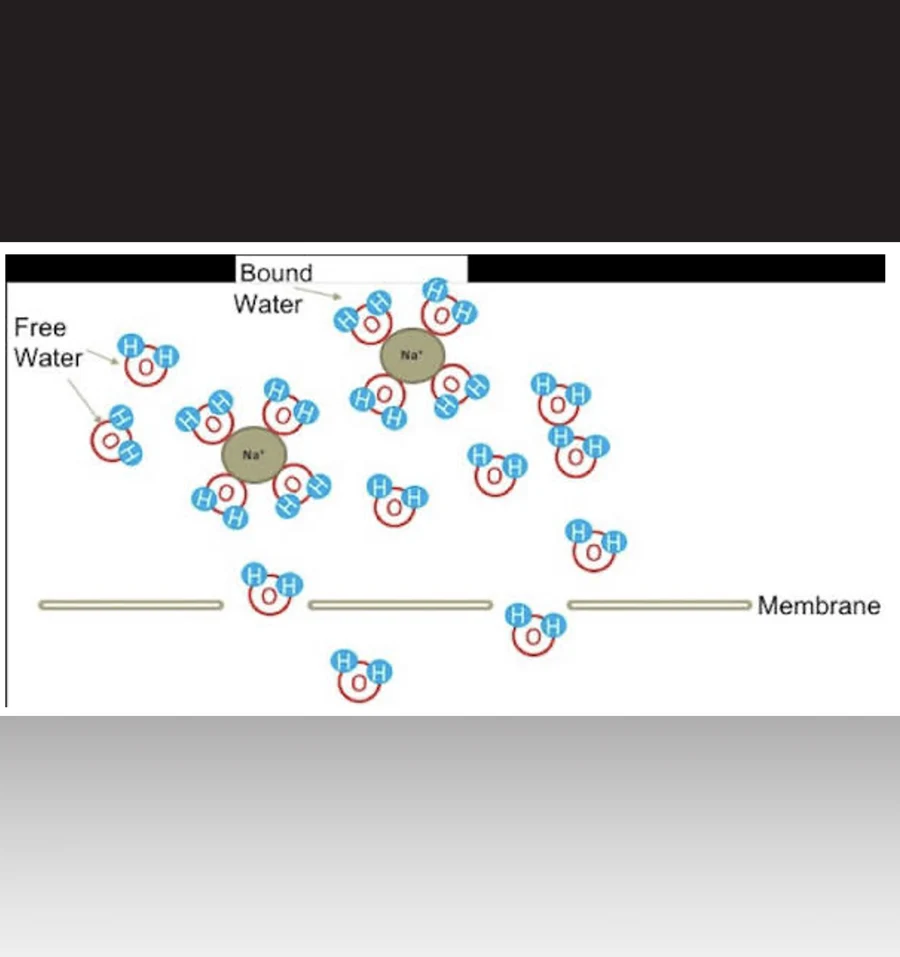
Free water (unbound) in tissue is mobile, fills extracellular/intracellular spaces, and acts as a solvent, while bonded (bound) water is tightly held by macromolecules (proteins, polysaccharides) through hydrogen bonding, acting as a plasticizer for structure. Free water is easily removed/frozen, whereas bound water resists removal, has a lower freezing point, and acts as a structural component.
After fixation in histology tissue processing, comes the second step; dehydration. Water has to be removed from the tissue to render it miscible with paraffin, to make blocks that can be cut on the microtome and then stained
The most used dehydration reagent is ethanol, pure or denatured. Both act similarly and you can refer to another article in the knowledge base about this subject https://chaptec.com/ca/articles/20250221105654/the-question-that-shocks.html
Ethanol is excellent; however, it will sometimes remove too much water from the tissue. How is this possible?
Water is tissue is either free or bound. Removing free water is what one wants to achieve for tissue processing. However, removing bound water renders the tissue very hard and brittle, making it difficult to cut as it has lost a component of its structure.
This is especially true for small biopsies. Because of their small size, they are submitted proportionally to more dehydration than a larger tissue for the same time of exposure.
This results in biopsies being difficult to section and tissue sometimes turning to dust while trying to cut them on the microtome. Experienced technologists will tell you that they have experimented difficulties cutting biopsies and they may not know exactly why.
New dehydrating reagents are offered that are made of methanol and isopropanol. The solvent characteristics of IPA also contribute to an improved “clearing”
Clearing is the second step of tissue processing. After dehydration is completed, the tissue is immersed in a solvent that will dissolve the fat in the tissues. These empty spots will later be filled with paraffin. It also makes the tissue transparent by bringing the refractive index close to the one of glass. This is usually performed with a solvent like xylene or toluene.
Extraction of lipids is a challenge for very fatty tissue like breast or brain. Using this new reagent will kick start the lipid extraction process simultaneously with dehydration, ensuring a better clarified tissue after the xylene step.
Incomplete lipid extraction may also lead to extremely difficult sectioning because of the remaining lipids (try and make a thin slice of butter!) If you experience this while sectioning, you could consider switching your dehydrating reagent.
Ethanol is also used in the staining process, as a destaining step after eosin in the H&E stain. The new dehydrating reagent has demonstrated that it works beautifully as an ethanol substitute for this application, rendering slides brilliant and higher in contrast.
Chaptec has launched Hydrofix our new dehydration reagent. Made in Canada, inexpensive and shipped rapidly. If you are encountering any of the above described challenges, Hydrofix could become an option and samples are available for evaluation Offered in all usual concentrations, 50, 70, 80, 90, 95 and 100%
What is the step that follows fixation in histology?
The next step is dehydration, which involves removing water from the tissue to allow it to be embedded in paraffin.
Why is it necessary to remove water from the tissue?
To make the tissue compatible with paraffin, so that blocks can be formed that can be cut on a microtome and stained.
What is the most commonly used reagent for dehydration?
Ethanol, either pure or denatured.
What problem can ethanol cause during dehydration?
It can remove too much water from the tissue.
Why is removing too much water problematic?
Because it also removes bound water, making the tissue hard and brittle, and therefore difficult to cut.
What are the two types of water present in tissues?
Free water and bound water.
What is free water?
It is mobile water that fills intra- and extracellular spaces and acts as a solvent.
What is bound water?
It is water attached to macromolecules (such as proteins), playing a structural role.
What is the difference between free water and bound water during dehydration?
Free water is easily removed, while bound water resists removal and is important for tissue structure.
Why are small biopsies more sensitive to dehydration?
Because they undergo proportionally more dehydration for the same exposure time.
What problem can arise when cutting biopsies?
They can become difficult to section or disintegrate into dust.
What is the step that follows dehydration?
“Clearing.”
What is the purpose of clearing?
To dissolve lipids and make the tissue transparent to facilitate paraffin infiltration.
What solvents are used for clearing?
Xylene or toluene.
Why can lipid extraction be difficult?
Because certain tissues, such as breast or brain tissue, are very high in fat.
What happens if lipids are not completely removed?
It makes cutting difficult, because the remaining lipids interfere with the section (like cutting through butter).
What new dehydration reagents are available?
Mixtures of methanol and isopropanol.
What is the advantage of isopropanol (IPA)?
It improves the clearing process and aids in lipid extraction.
Is ethanol used for anything other than dehydration?
Yes, in H&E staining as a decolorization step after eosin.
Can the new reagents replace ethanol?
Yes, they can serve as an effective substitute and improve slide contrast.
What product has Chaptec launched?
Hydrofix, a new dehydration reagent.
What are the advantages of Hydrofix?
It is cost-effective, made in Canada, shipped quickly, and available in several concentrations.
| 470, avenue Laurendeau, Montréal-Est (Quebec) H1B 5M2 | |||
| Phone: | 514 498-3620 | Toll free: | 833 498-3620 |
| Email: | chaptec@chaptec.com | ||

